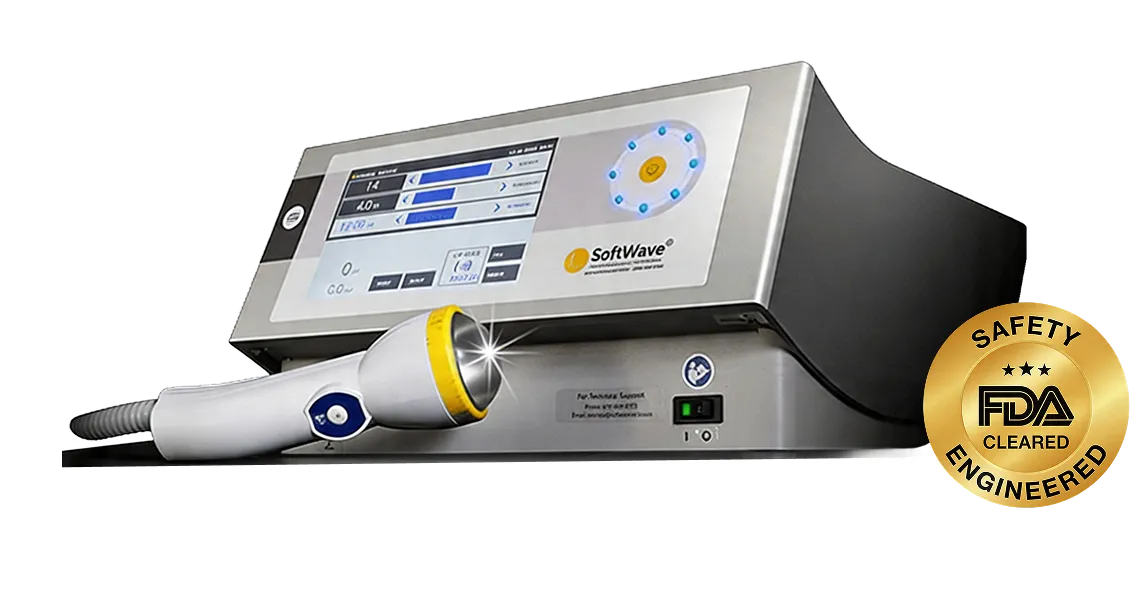
HEALTHCARE PROFESSIONAL USE ONLY. NOT FOR PATIENT DISTRIBUTION

The New Standard in Evidence-Based Podiatric Care
Optimize patient outcomes and practice revenue with the only FDA-cleared, broad-focused electrohydraulic shockwave therapy for chronic wounds and musculoskeletal pathologies.
SoftWave utilizes a patented electrohydraulic design to deliver a broad-focused shockwave that initiates a biological response without the tissue trauma associated with radial or focused devices.
Angiogenesis: Stimulates the expression of VEGF and PCNA to improve microcirculation and local perfusion.
Stem Cell Activation: Recruits and activates endogenous mesenchymal stem cells to accelerate the repair of degenerated tendons and ligaments.
Anti-Inflammatory Response: Modulates NF-kB pathways to significantly reduce pro-inflammatory cytokines.
Pathologies Addressed: Proven efficacy for Plantar Fasciitis, Achilles Tendinopathy, Morton’s Neuroma, and Peripheral Neuropathy.






Lorem Ipsum
is simply dummy text
Of the printing and typesetting industry. Lorem Ipsum has been the industry's
Become a provider today
Learn more about how SoftWave technology can improve your patients' outcomes.
Deep Tissue Propagation
SoftWave’s patented broad-focused applicator facilitates the distribution of acoustic energy across extensive superficial and deep tissue planes, eliciting a biological response that activates endogenous healing mechanisms within the targeted area.
FDA Cleared for:
Activation of connective tissue
Temporary Improved blood supply
Pain reduction
Treatment of chronic diabetic foot ulcers
Treatment of acute second-degree burns
Health Canada Licensed for:
Plantar Faciitis
Heel Spur
Achillodynia
Acute Wounds and Burns
Chronic Wounds and Ulcers
Propagation Into Tissue
SoftWave delivers deep tissue penetration, unlocking the full potential of biological effects for better healing.
Comprehensive Podiatric Indications
From chronic plantar fasciitis to complex wound care, provide your patients with the gold standard in electrohydraulic shockwave therapy.
Plantar Fasciitis
SoftWave Therapy can help manage pain and discomfort by promoting increased blood flow and tissue activation.
Achillodynia (Achilles Tendon Pain)
The therapy aids in reducing inflammation and supporting the repair of damaged tendons, helping alleviate symptoms.
Diabetic Foot Ulcers (DFUs)
SoftWave’s ability to enhance circulation and stimulate tissue can be effective in managing symptoms associated with chronic wounds.
Heel Spur
By addressing inflammation and improving blood supply, SoftWave Therapy can help reduce discomfort related to heel spurs.
Morton's Neuroma
Symptoms of pain and inflammation may be reduced through the benefits of SoftWave Therapy.
Peripheral Neuropathy
SoftWave Therapy can enhance nerve function and help manage symptoms related to nerve damage, especially in diabetic patients.
Chronic Wounds
The increased circulation from SoftWave Therapy can support the management of chronic wounds and aid in the healing process.
Bunion Pain
While not a curative treatment for bunions, SoftWave Therapy may help manage pain and discomfort by reducing inflammation.
62-Year-Old DFU Patient Before &
After 4 SoftWave Sessions
Advanced Tissue Regeneration for Complex DFUs
Replace high-cost, time-consuming methods with SoftWave Therapy—the only electrohydraulic shockwave FDA-cleared for the treatment of chronic diabetic foot ulcers.
See how SoftWave’s electrohydraulic technology activates the body’s natural healing response to close stubborn wounds in just four sessions.

Advanced Tissue Regeneration for Complex DFUs
Replace high-cost, time-consuming methods with SoftWave Therapy—the only electrohydraulic shockwave FDA-cleared for the treatment of chronic diabetic foot ulcers.
See how SoftWave’s electrohydraulic technology activates the body’s natural healing response to close stubborn wounds in just four sessions.


It helps our patients heal faster
Having one machine that does so much— without the need for steroids—is incredible. It helps our patients heal faster, improves their quality of life, and boosts our clinic’s productivity. It’s a great investment and has become a powerful referral source.
– Dr. Benjamin Weaver, DPM, FACFAOM, FAPWCA, CWS, FCCWS, FCND, DAPBOPPM, CMET
Founder of Central Kansas Podiatry Associates

Clinical Studies Supporting SoftWave Therapy

Plantar Fasciitis
Efficacy of Unfocused Medium-Intensity Extracorporeal Shock Wave Therapy (MI-ESWT) for Plantar Fasciitis.

Achilles Tendiopathy
Current evidence of extracorporeal shock wave therapy in chronic Achilles tendinopathy

Heel Pain
American College of Foot and Ankle Surgeons Clinical Consensus Statement: Diagnosis and Treatment of Adult Acquired Infracalcaneal Heel Pain

Sports Medicine
Extracorporeal Shockwave Therapy in the Management of Sports Medicine Injuries
Lorem Ipsum is
Lorem Ipsum is simply dummy text of the printing and typesetting
since
Lorem Ipsum is simply
Lorem Ipsum is simply dummy text of the printing
Lorem Ipsum
Lorem Ipsum is simply dummy text of the printing and typesetting since
the
Lorem Ipsum
Lorem Ipsum is simply dummy text of the printing and typesetting industry
since the
Declining Reimbursement
Successful Providers are staying ahead in a declining reimbursement environment by adopting SoftWave’s non-invasive solution as their hybrid solution.
Clinical Regulatory & Results
FDA 510(k) Cleared Technology
Increased Circulation: Proven to enhance local blood flow to treated areas.
Tissue Activation: Promotes the activation of connective tissue.
Advanced Healing: Indicated for the treatment of chronic diabetic foot ulcers and second-degree burns.
Pain Management: Non-invasive relief of minor muscle aches and pains.
Science & Trust
Evidence-Based Care
Clinical Integrity: Supported by extensive peer-reviewed clinical research.
Data-Driven Results: We partner with Databiologics to provide real-world data tracking.
Enhanced Trust: Move from anecdotal claims to data-driven consultations to increase patient satisfaction and case acceptance.
Authority & Elite Use
Trusted by Medical Centers and Top Sport Team
Global Recognition: Utilized at leading national and international research and medical centers.
The Choice of Champions: SoftWave is the preferred technology for professional organizations across the MLB, NFL, NBA, NHL, MLS, and PGA.
Clinical Standard: Trusted by the UFC, WNBA, and Premier League for rapid recovery.
Get the Revenue Growth Blueprint
See how SoftWave adds $10k–$30k in monthly cash-pay revenue.

This page is intended for licensed healthcare professionals only and is not directed at patients or consumers. SoftWave TRT holds FDA 510(k) clearance for activation of connective tissue, treatment of chronic diabetic foot ulcers, treatment of acute second-degree burns, and improved blood supply. Individual patient outcomes vary and are not guaranteed. Financial results are based on reported practice usage and are not a guarantee of income or return on investment. Results depend on individual practice size, patient volume, session pricing, and clinical use. SoftWave TRT is not intended to diagnose, treat, cure, or prevent any disease beyond its FDA-cleared indications.